Prevention and Key Points
Feline Factsheet
Prevention
In endemic areas, general prevention of sand fly bites is based on the same procedures as for dogs.
It is advised to use topical insecticides currently licensed for cats to protect:
| Individual cats and reduce their risk of developing infection and clinical disease. Feline populations in endemic areas (particularly infected cats) to improve the control of L. infantum infection in the vector. |
The majority of insecticides currently available for cats are not safe or effective in preventing the bites of sand flies.
Collars releasing a combination of flumethrin and imidacloprid are the only pyrethroid formulation product licensed for cats and proven to reduce the incidence of L. infantum infection in a field study.
Testing blood donors (antibody detection and blood PCR) reduces the potential risk for non-vectorial transmission via blood transfusion.

Key Points
| Leishmania infantum is most likely transmitted to cats by sand flies. There is currently no demonstration of nonvectorial transmission in cats as described in dogs, although transmission by blood transfusion may occur. The prevalence of L. infantum infection in cats is commonly lower than that of canine infection in endemic areas, but it is not negligible. Cats seem to be less susceptible than dogs to L. infantum infection. Sub-clinical feline infections are common in areas endemic for CanL, but clinical disease appears to be less common in cats. Skin and mucocutaneous erosive or nodular lesions, lymph node enlargement and hypergammaglobulinemia are the most common clinical and clinicopathological findings, followed by ocular and oral lesions, proteinuria and mild non-regenerative anemia. Infected cats can represent an additional domestic reservoir for L. infantum infection. Diagnosis is based initially on cytological and quantitative serological techniques in cats with clinical signs and/or clinicopathological abnormalities compatible with disease. Rapid tests validated to detect anti-L. infantum antibodies in cats are currently not available. Currently, treatment is empirically based on some drugs used also for dogs. Most pyrethroids are toxic for cats and only collars releasing a combination of flumethrin and imidacloprid are currently licensed and proven as safe for use in the prevention of L. infantum infection in cats. |
Monitoring and Prognosis
Feline Factsheet
Relapse of disease is frequently observed after the end of anti-Leishmania treatment. Clinical signs and/or clinicopathological abnormalities can be different from those manifested previously. Careful monitoring should include physical examination, CBC, biochemical profile, urinalysis with urinary protein:creatinine ratio (UPC) and quantitative serology at the frequencies indicated below (Table 3).
Available information on the progression of FeL cases is still limited compared to knowledge on the disease in dogs, but the life expectancy of cats with leishmaniosis seems to be good (years, even in untreated and FIV positive cats). However, concurrent conditions (malignant neoplasia or FIV/FeLV infections among others) or complications (particularly renal disease) occur and should be considered when estimating the patient’s prognosis.
Table 3. Monitoring of cats diagnosed with FeL
| ACTION | FREQUENCY |
| Physical examination Complete blood count Serum biochemical profile Urinalysis including UPC | At least fortnightly during the first month of therapy and then at least every 3 months in the first year or after stopping therapy Thereafter every 6 months in cats clinically recovered |
| Quantitative serology | Every 3-6 months during the first year or after stopping therapy Every 6 months after the first year |
Therapy
Feline Factsheet

Information regarding the therapy for FeL is currently derived solely from descriptive, non-controlled studies.
Empirical treatment involving two of the most recommended drugs for dogs is generally considered clinically effective and appears to be safe in most cases. The most frequently used protocol is currently the combination of meglumine antimoniate and allopurinol. However, allopurinol or meglumine antimoniate monotherapies have also been found clinically effective and widely used, particularly in case of adverse effects with one of the two drugs. In certain instances, allopurinol is the preferred choice when owners prefer to avoid performing subcutaneous injections.
Table 2. Drugs most frequently used for treating FeL (currently used in combination)
| Drugs | Dose | Adverse effects |
| Meglumine antimoniate | 50 mg/kg SC, q24h for 30 days | Acute kidney injury |
| Allopurinol | 10 mg/kg PO q12h or 20 mg/kg PO q24h for at least 6-12 months | Acute kidney injury Head and neck dermatitis/Pruritus Increased liver enzymes Xanthinuria |
PO: Per os – SC: Subcutaneous
Cats under therapy with allopurinol and/or meglumine antimoniate should always be carefully monitored for any adverse effects. Suspected acute kidney injury (meglumine antimoniate and/or allopurinol) and reversible skin reactions (allopurinol) were reported in some cases.
Propylene glycol is among the excipients of the miltefosine oral formulation licensed for the treatment of CanL.
Propylene glycol can cause Heinz body formation in feline red blood cells as demonstrated in a pilot study on healthy cats treated with Milteforan© for 15 days. A consequence of Heinz body formation is anemia, due to a decreased life span of feline red blood cells. Therefore, miltefosine is not recommended for the treatment of FeL.
Disclaimer: Information given here on drugs and dosages are based on a consensus of clinical and scientific experience by the LeishVet members. Most recommendations have been published in scientific peer-reviewed journals. Veterinary practitioners are requested to check with product leaflets and product registrations in their related country prior to any product selection and initiation of treatment.
Clinically Healthy Infected Cats
Feline Factsheet
Clinically healthy infected cats are antibody-positive (and/or PCR-positive) cats with no clinical signs and clinicopathological abnormalities compatible with FeL. These cats may have coinfections or other diseases, and it is recommended to follow up L. infantum infection considering their risk to develop FeL (Figure 11).
Figure 16. Management of cats with serological and/or PCR-positive positivity for L. infantum and no clinical signs and clinicopathological abnormalities compatible with FeL (clinically healthy infected cats)

BM: Bone marrow – FeLV: Feline leukemia virus – FIV: Feline immunodeficiency virus – LN: Lymph node
A cat will be considered with leishmaniosis if histopathological findings are compatible with leishmaniosis and IHC or PCR are positive.
It is not common to visualize amastigotes in histology and therefore, IHC is always recommended. If IHC is negative, PCR is also recommended.
Purposes of Diagnosis
Feline Factsheet
1. Confirm the etiology of disease in cats with clinical signs and/or clinicopathological abnormalities compatible with FeL (Figure 15)
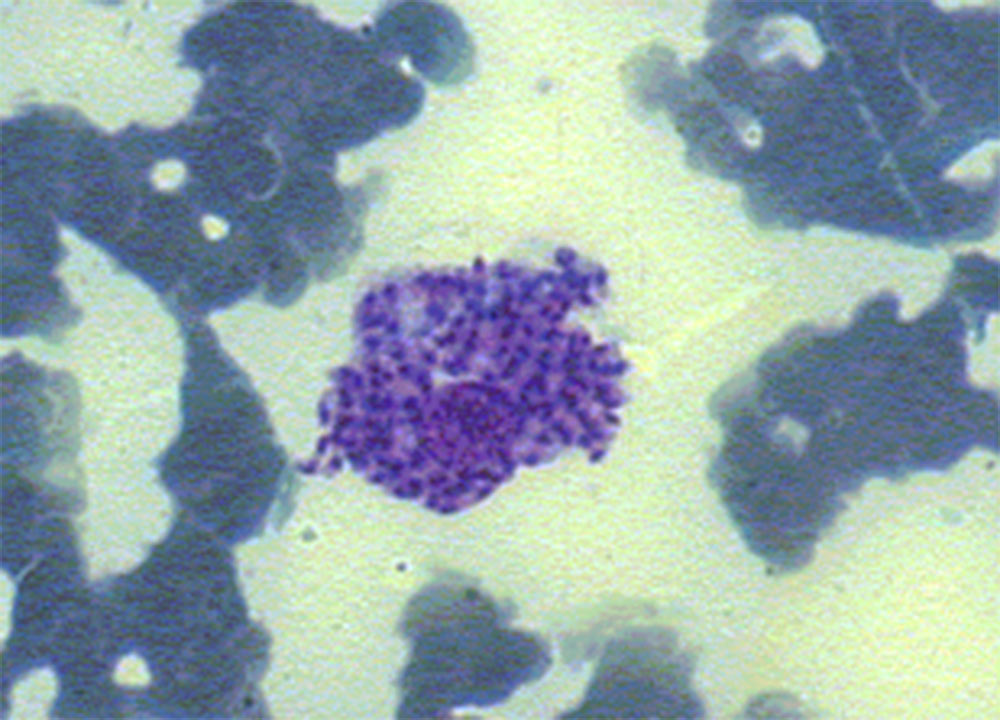
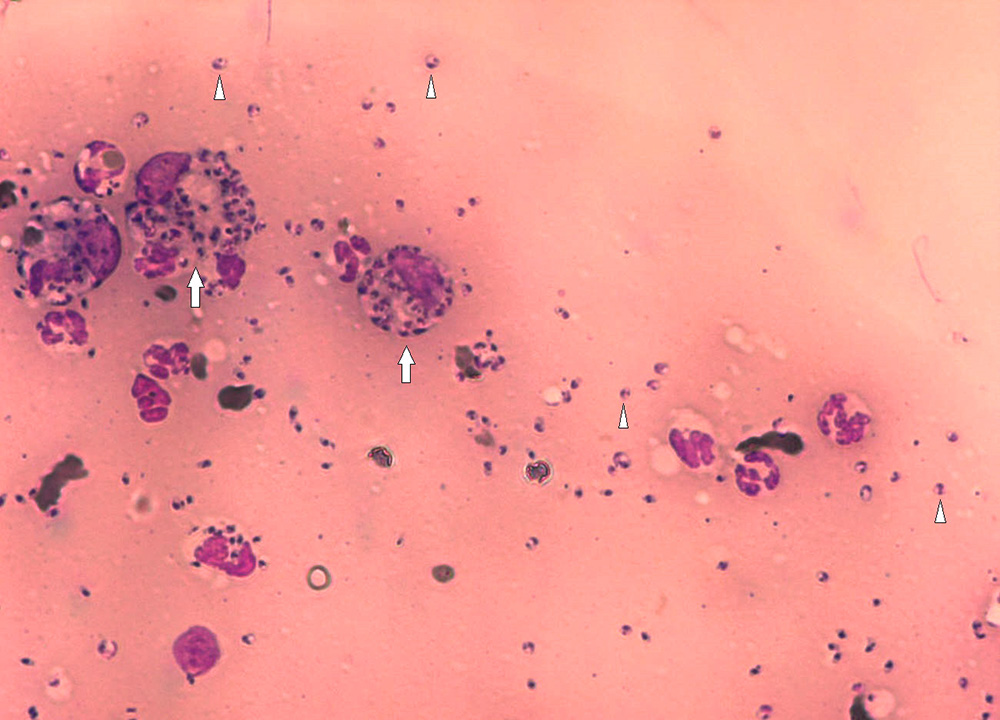
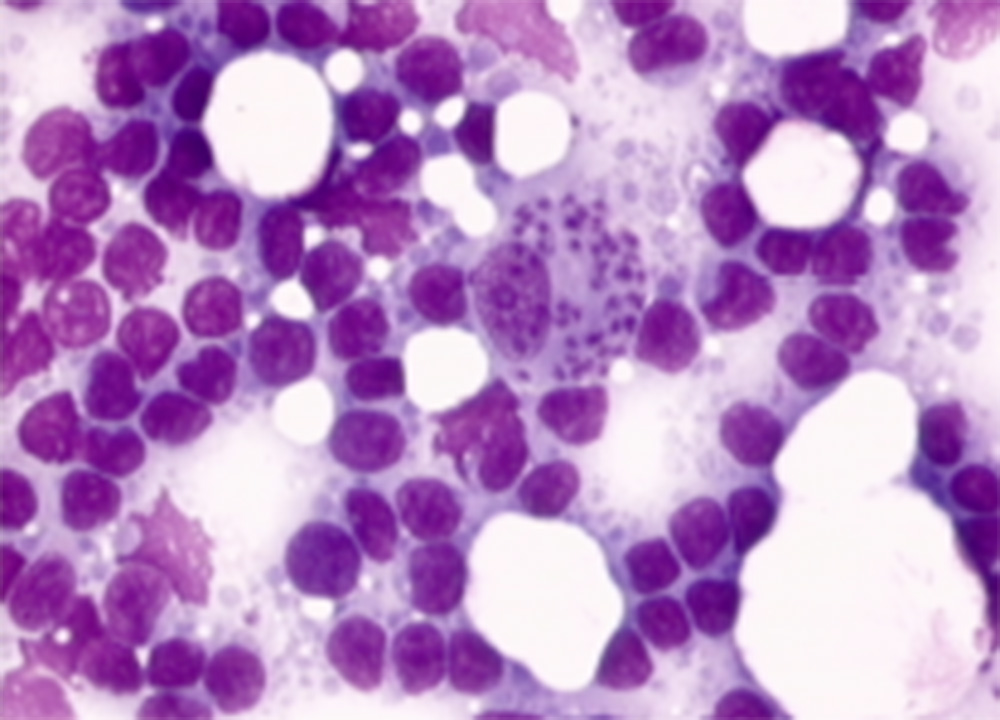
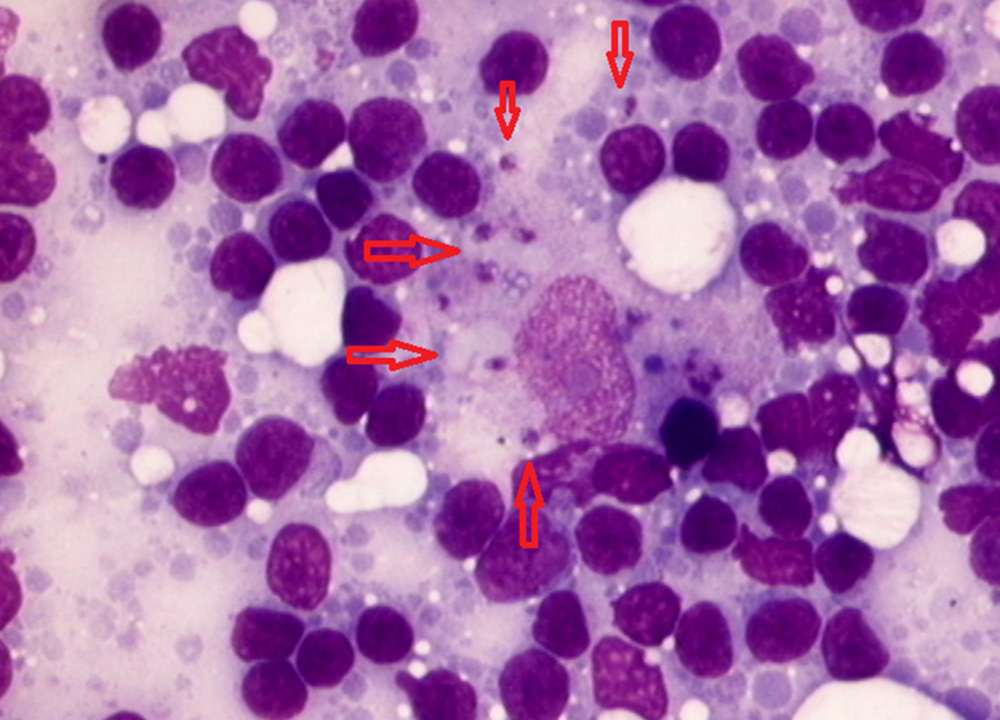
High levels of antibody positivity (i.e. in case of IFAT > 3 dilutions over the validated cut-off value) detected by quantitative serology (IFAT, ELISA or DAT) confirm diagnosis in cats with a risk of exposure to L. infantum infection and suggestive clinical signs and/or clinicopathological abnormalities (Table 1). However, cytological evaluation of any skin, mucosal, or mucocutaneous lesions, enlarged lymph nodes or spleen, or any other lesion evidenced by imaging and endoscopy should be performed. Compatible cytological findings (such as lymphoid hyperplasia in lymphoid organs; and neutrophilic and/or macrophagic as well as lymphoplasmocellular inflammation) and/or visualization of amastigotes is suggestive or confirmatory of L. infantum infection, respectively (Figures 11-14). Cytology also permits to detect comorbidities. Histopathological evaluation and/or Leishmania immunohistochemistry may be required based on each patient’s list of problems.
In antibody-negative or low-positive cats with no evidenced tissue lesions, Leishmania amastigotes (cytology evaluation) or parasite DNA (PCR) can be detected in bone marrow, lymph nodes or spleen. However, as comorbidities are possible, they also must be investigated based on the individual list of problems, to confirm a causative role of L. infantum infection in the clinical presentation (i.e. differentiation of infection from disease).
Point-of-care tests validated to detect anti-L. infantum antibodies in cats are currently not available.
2. Screening of apparently healthy cats living or travelling to or from endemic areas
The screening of clinically healthy cats by means of quantitative serology and PCR techniques is recommended in endemic areas in case of the following conditions: blood donors, cats requiring chronic immunosuppressive therapies, and before relocating cats to non-endemic areas. Imported/relocated cats must be tested at arrival, but seroconversion can take months and retesting should be considered six months later, in case of presenting compatible clinical signs, before any immunosuppressive therapy or becoming a blood donor. In case of healthy individuals, generally minimally invasive tissue sampling is preferred (blood, lymph nodes and conjunctival swabs) for PCR, however their sensitivity is low and it should be increased by performing multiple tissue PCR testing.
Figure 15. Flow chart for the diagnostic approach to cats with clinical signs compatible with FeL
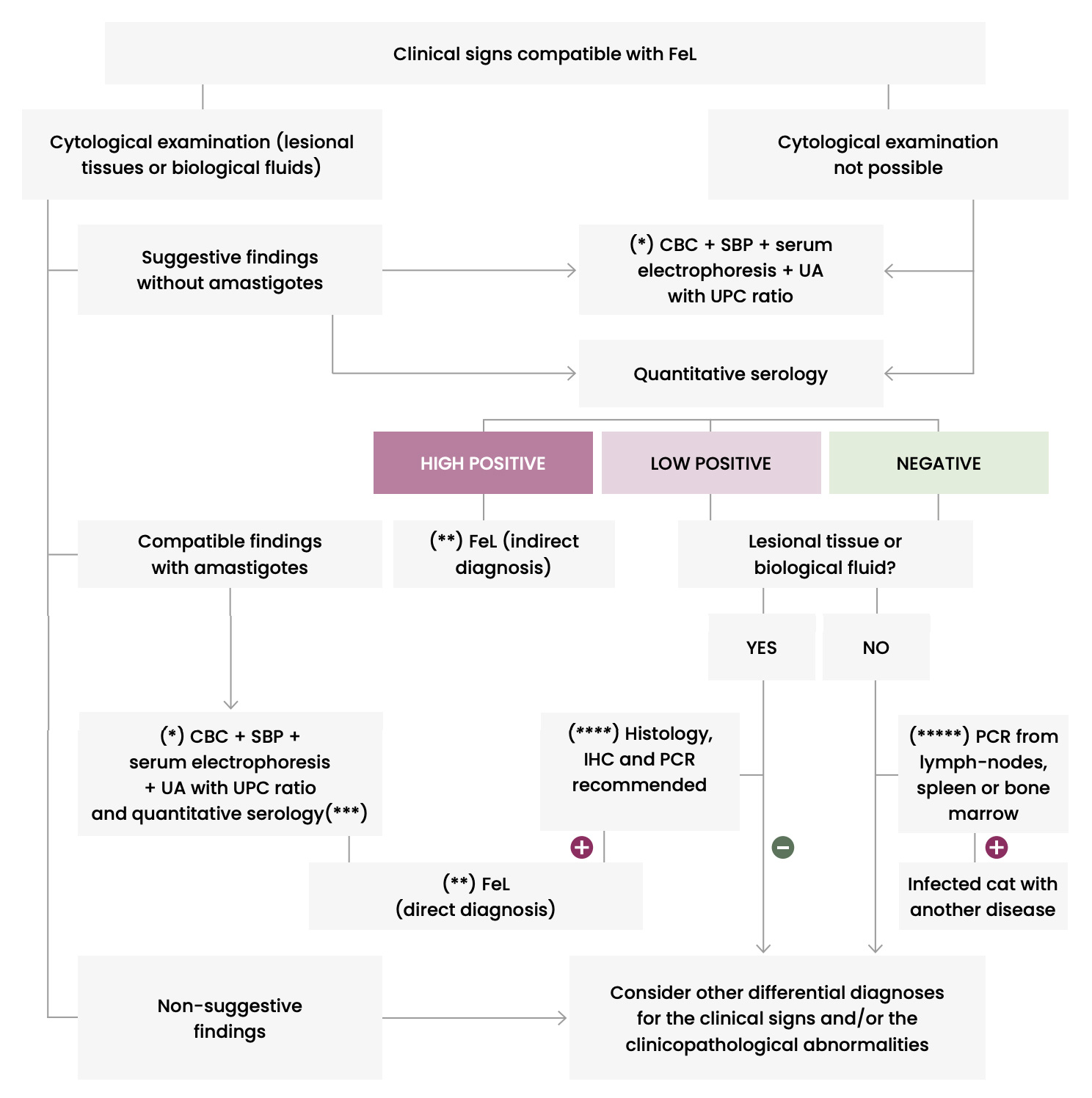
CBC: Complete blood count – IHC: Immunohistochemistry – PCR: Polymerase chain reaction – SBP: Serum biochemical profile – UA: Urinalysis – UPC: Urinary protein:creatinine ratio
(*) To better check health status and evaluate clinicopathological abnormalities suggestive of FeL.
(**) Conventional anti-Leishmania treatment and monitoring for resolution of clinical signs and clinicopathological abnormalities.
(***) Quantitative serology is recommended for the post-treatment follow-up.
(****) A cat will be considered with leishmaniosis if histopathological findings are compatible with leishmaniosis and IHC or PCR are positive. It is not common to visualize amastigotes in histology and therefore, IHC is always recommended. If IHC is negative, PCR is performed to confirm or rule out infection.
(*****) Detection of infection based on clinical suspicion.
Leishmaniosis in Cats
Feline Factsheet
Most FeL case reports are from European and Mediterranean endemic areas where the number of pet cats is high. However, FeL remains rare, as a disease, even in areas where leishmaniosis is common in dogs and feline infection is frequent. It is postulated that cats are therefore less susceptible than dogs to L. infantum infection. However, it cannot be excluded that the disease is still underdiagnosed because it is neglected – or even ignored – by many practitioners, and clinical suspicion is challenging when suggestive signs are lacking, or the clinical presentation is overlooked by signs of a concurrent disease. Moreover, in some endemic areas, the level of cat medical care is generally lower compared to dogs.
About 140 clinical cases have been reported in Europe during the last 3 decades (Italy, France, Spain and Portugal) with some cases diagnosed in Germany, Switzerland and the UK in cats imported from endemic regions.
Host factors predisposing to susceptibility probably exist, as roughly half of the reported clinical cases have been observed in cats that could have had an impaired immune system secondary to FIV or feline leukemia virus (FeLV) or dual FIV and FeLV coinfections, long-term immune-suppressive therapies, malignant neoplasia, or debilitating diseases. However, the level of evidence of this observation is low as it is not supported by controlled studies.
The disease has been diagnosed in adult cats (2-21 years) with a median age of 9 years.
The incubation period can last years as suggested by clinical manifestations observed in non-endemic areas several years after the relocation of cats from endemic areas.
The course of disease is mostly chronic and progressive.
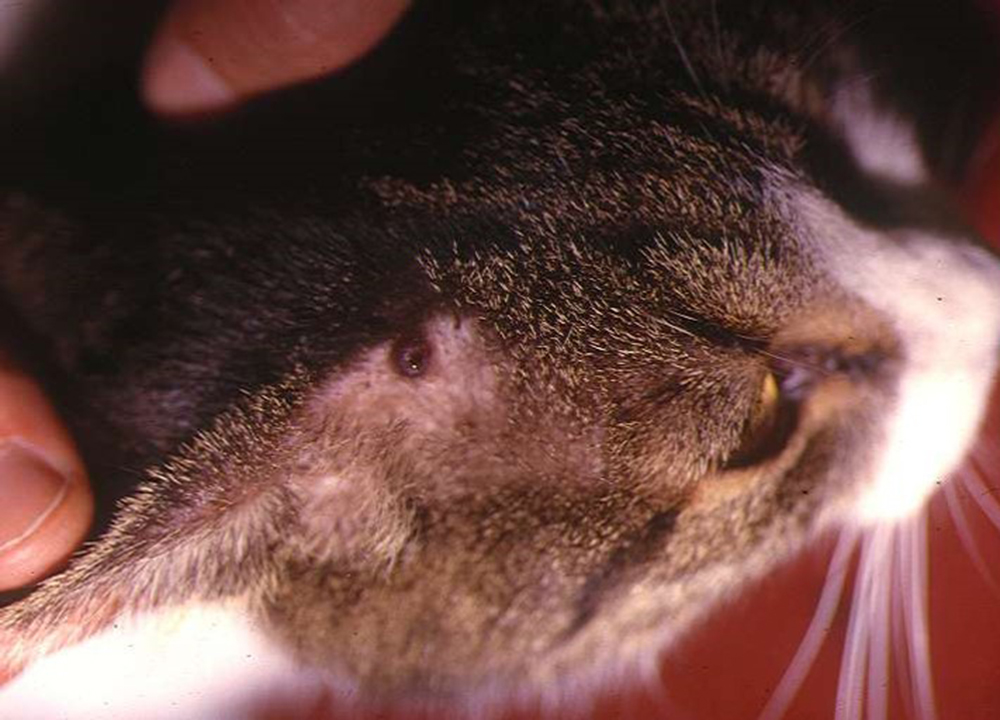
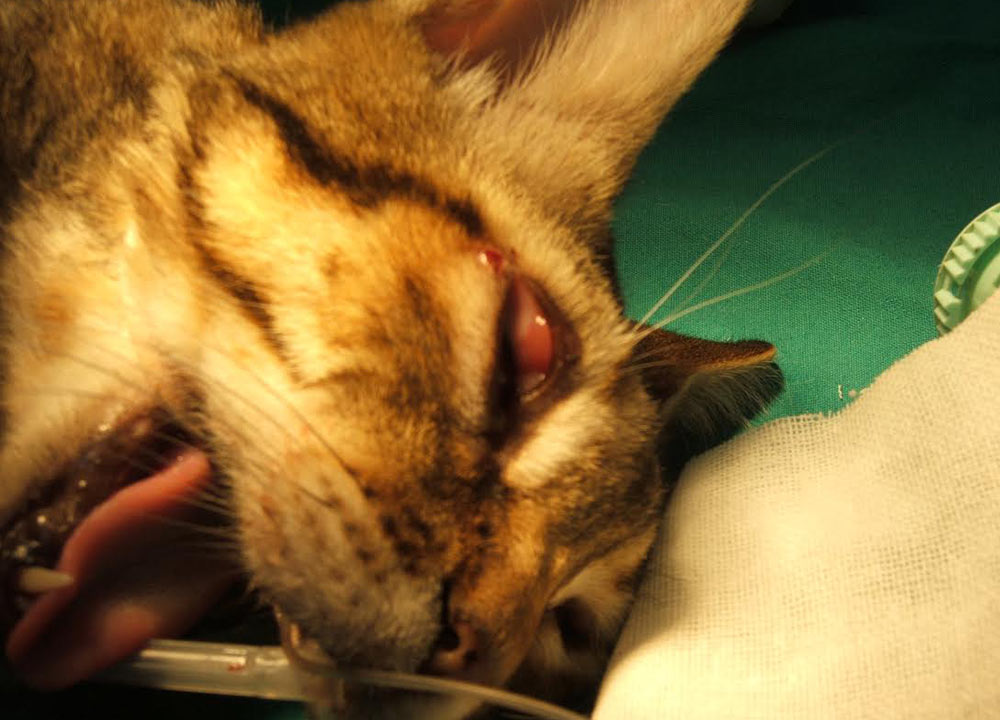
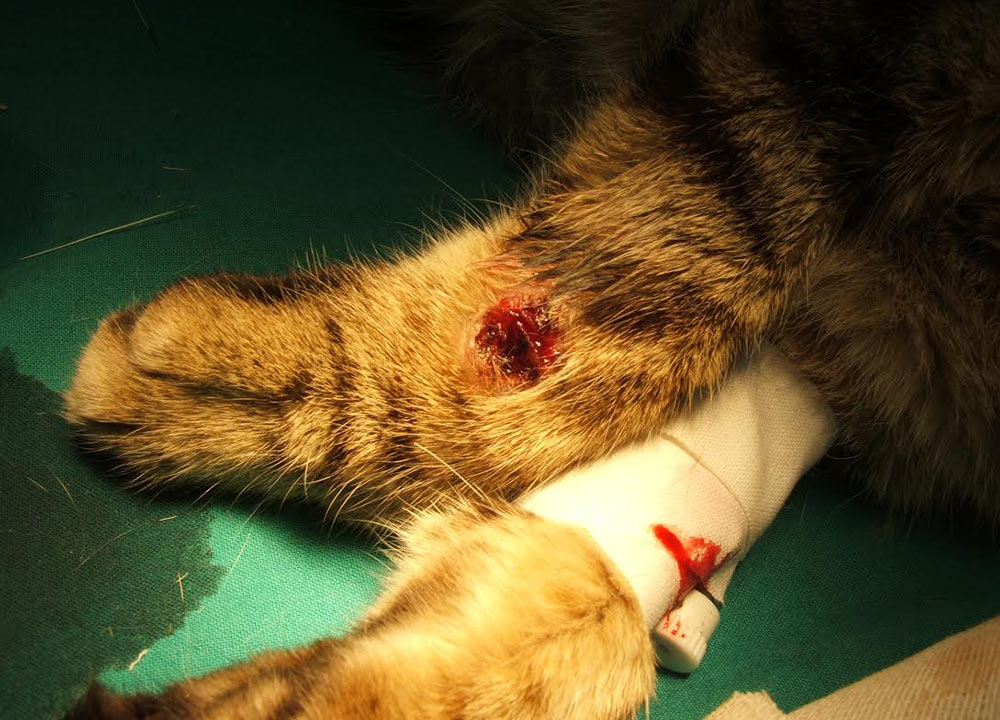
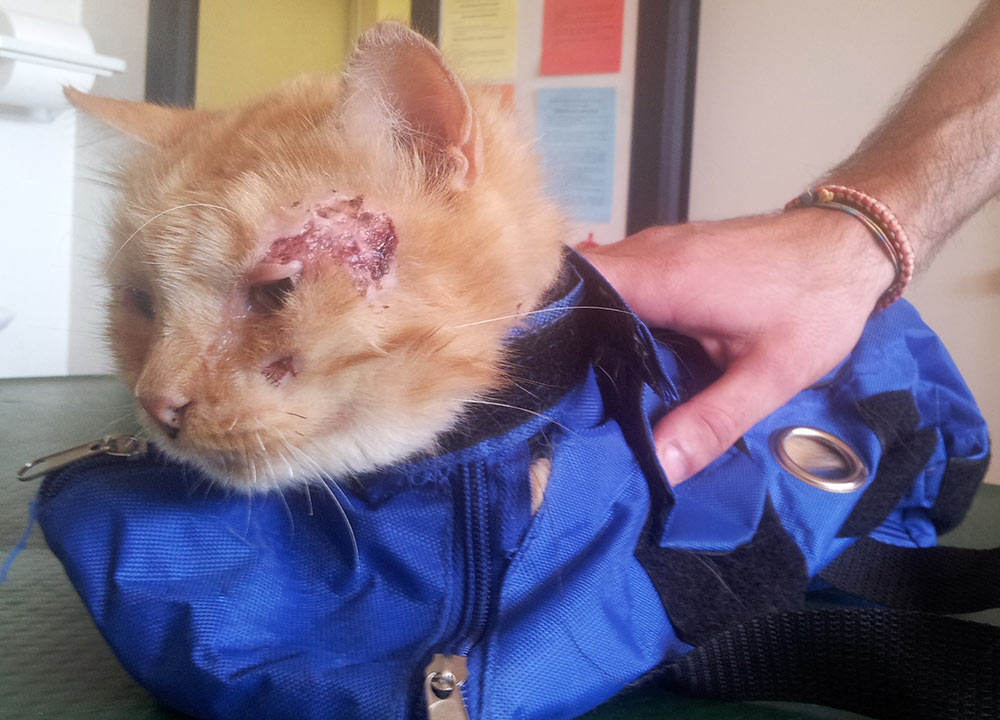
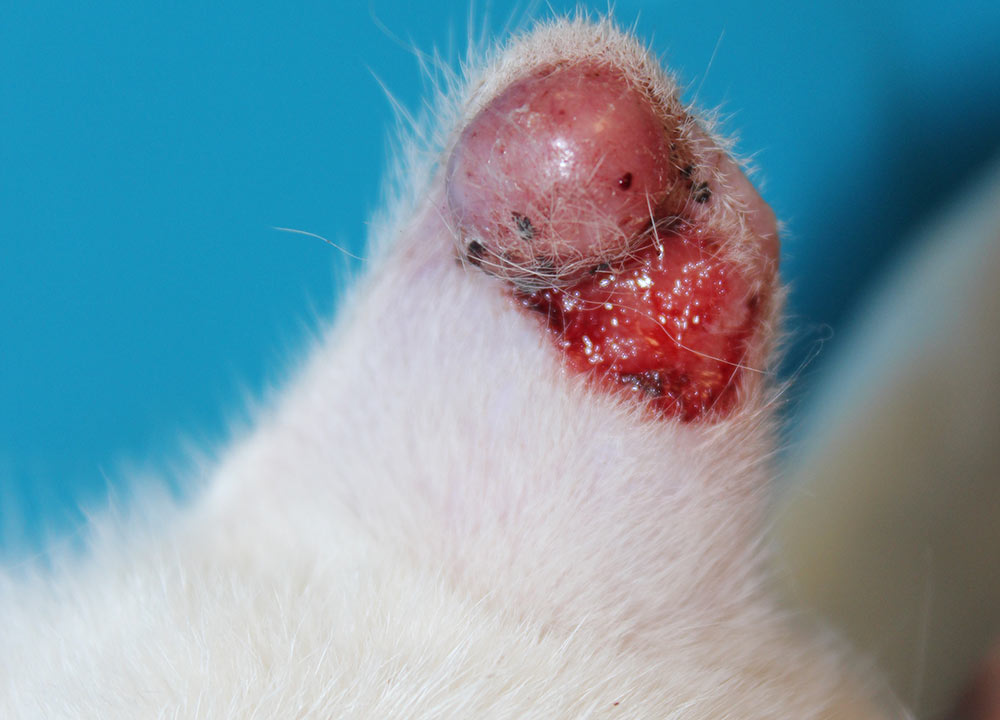
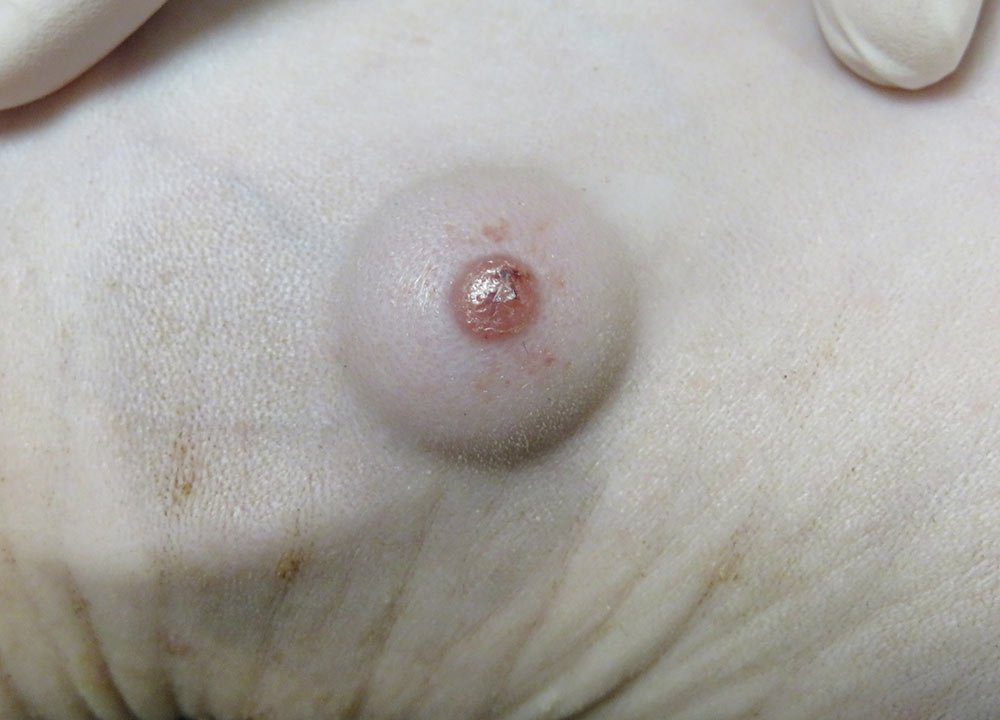
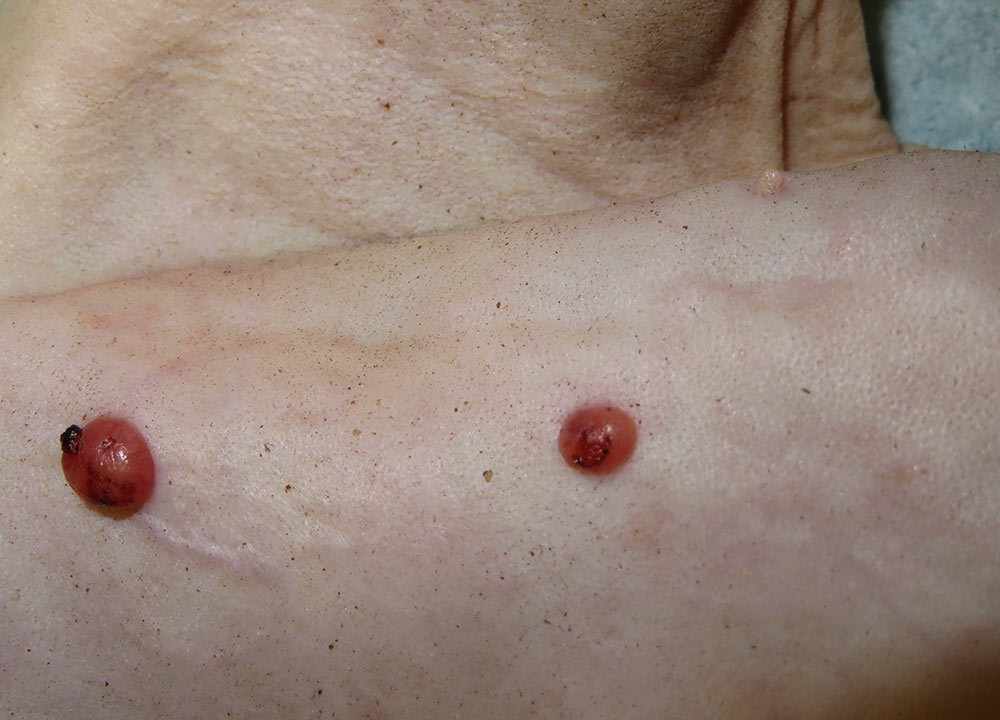
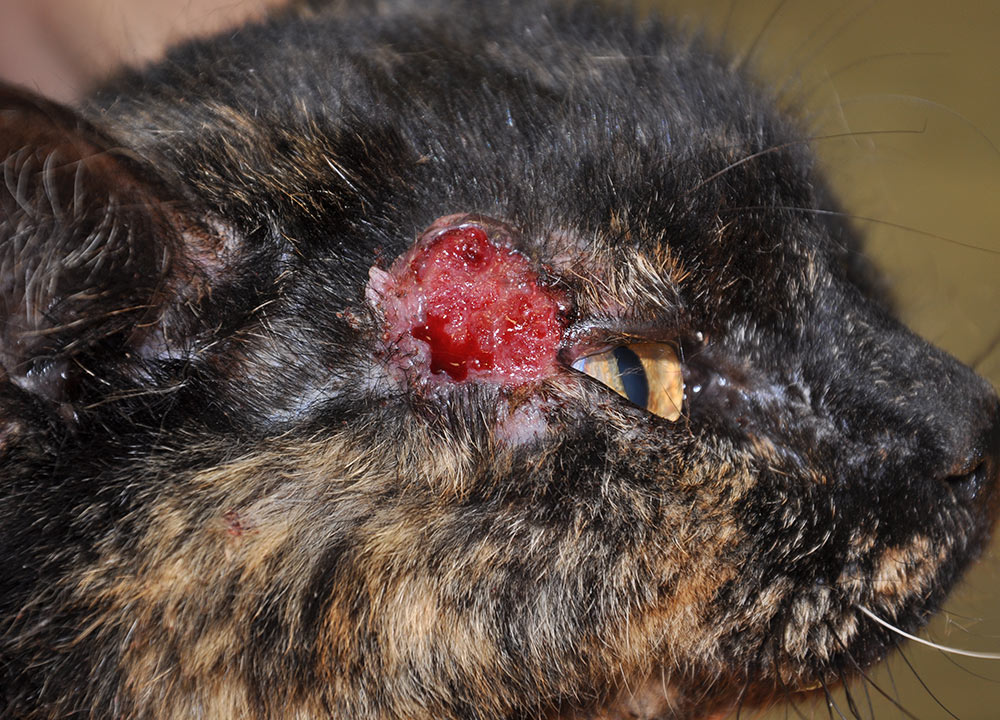
Similar to canine leishmaniosis, cutaneous and mucocutaneous lesions and lymphadenomegaly are the most frequently reported manifestations (Table 1). Ulcers and nodules are the most common cutaneous and mucocutaneous lesions observed, mainly distributed on the head or on distal limbs (Figures 2-8). Hemorrhagic nodules are atypical skin lesions (Figure 9).
Uveitis is a frequent ocular lesion (Figure 10) in FeL. Oral lesions consist of nodules (tongue and gingival mucosa).
In recent years, atypical presentations have been reported including upper respiratory tract disease manifested with chronic nasal discharge and severe obstructive syndrome in case of granulomatous inflammation with nodular or infiltrative masses. Other single cases with atypical presentations where the causative role of L. infantum has been documented with presentations of chronic diarrhea, mastitis and cutaneous panniculitis.
The clinicopathological evaluation of cats with leishmaniosis shows hyperglobulinemia with hypergammaglobulinemia frequently (Figure 10), mild normocytic normochromic non-regenerative anemia, and proteinuria.
Pictures: © Laura Ordeix and Maria Grazia Pennisi
Table 1. Clinical and clinicopathological abnormalities reported in FeL (listed in decreasing order of frequency)
| CLINICAL FINDINGS | CLINICOPATHOLOGICAL ABNORMALITIES |
| Frequent* | |
|
Skin and/or mucocutaneous lesions Lymphadenomegaly |
Hyperglobulinemia Hypergammaglobulinemia |
| Uncommon** | |
|
Ocular lesions Oral lesions General signs (lethargy, anorexia,weight loss) |
Proteinuria Mild-to-moderate non-regenerative anemia |
| Rare*** | |
|
Pale/icteric mucous membranes Splenomegaly and/or hepatomegaly Chronic nasal discharge Upper airway obstructive syndrome Cachexia Polyuria/Polydipsia Fever Diarrhea Mastitis Subcutaneous panniculitis Abortion |
Azotemia Hypoalbuminemia Monocytosis Neutrophilia Pancytopenia |
* around 50% of cases
** around 30% of cases
*** less than 25% of cases
Prevalence of Infection and Coinfections
Feline Factsheet

Most information regarding feline L. infantum infection has come from investigations performed in the Mediterranean Basin and the Middle East.
The prevalence of L. infantum infection in cats, as evaluated in studies performed in many countries of the Mediterranean Basin and the Middle East (Albania, Algeria, Cyprus, Egypt, France, Germany, Greece, Iran, Israel, Italy, Portugal, Qatar, Spain, Turkey) from the 1980’s is variable. Both antibody and blood molecular prevalences have been investigated with diverse serological and PCR techniques in populations of cats with different characteristics and in countries with different socio-economical situations, and this can contribute to the wide range of positivity observed (seropositivity: 0-75.0%; blood PCR positivity: 0-60.7%). However, prevalence of Leishmania infection in cats is commonly lower than the prevalence in dogs in the same area.
Some studies have found that coinfections with other pathogens are detected in Leishmania-infected cats. However, this finding can be accidental and mostly due to a high prevalence of the co-infecting microorganisms in cats (e.g.: Bartonella, Toxoplasma). Conversely, there is evidence that feline immunodeficiency virus (FIV) seropositive cats have a higher risk of being affected by L. infantum infection.
Etiology, Geographic Distribution and Transmission
Feline Factsheet
Diseases caused by Leishmania spp. in cats are far less known than in dogs and humans, and felines had been considered to be less susceptible than dogs to Leishmania infection in the past. However, a high number of investigations confirmed in the last decades the susceptibility of cats to Leishmania infection, and that they are infectious to sand fly vectors. Additionally, case reports have increasingly been reported, but controlled field studies about clinical management and therapy are still lacking.
From the above considerations it follows that feline leishmaniosis (FeL) is a topic of interest for feline medicine and that cats play a role in the One Heath approach of leishmaniosis.
Etiology, Geographic Distribution and Transmission
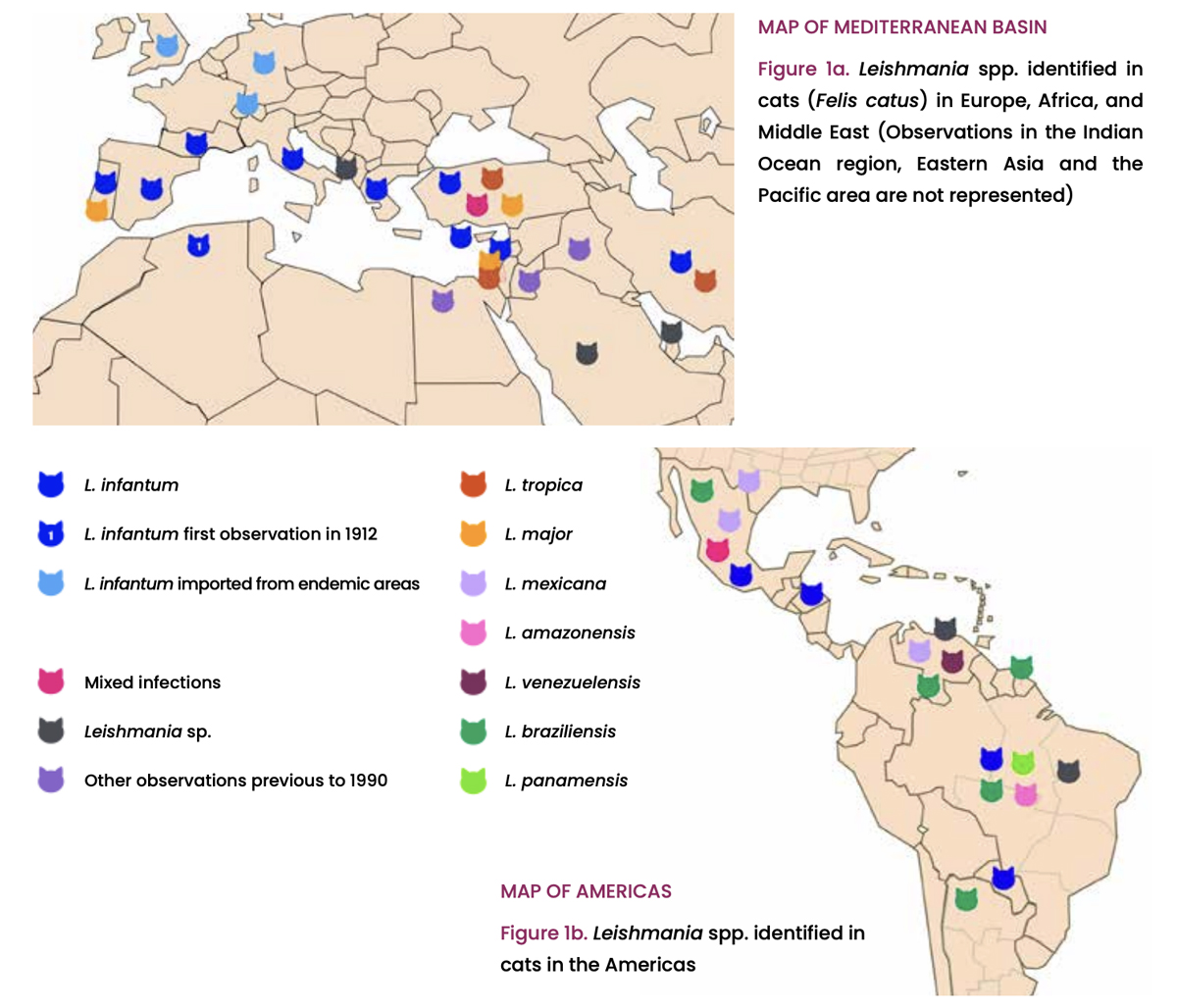
Cats are potentially infected by the same Leishmania spp. that infect dogs and humans in endemic areas all over the world (Figure 1). Leishmania infantum is the most frequently reported species infecting cats, and this fact sheet is based on published data on L. infantum infection and the associated feline disease, mostly reported in the Mediterranean Basin and Middle East region. However, this species is largely described in cats also in Central and South America.
Leishmania infantum is transmitted to cats by sand flies, as they have been shown to feed on cats and to be infected after feeding on naturally infected cats. Moreover, sand flies infected from cats transmitted the infection to dogs. Considering that infected cats are a source of infection to sand flies and generally suffer from chronic infection, LeishVet postulates that, based on new insights in the epidemiology of leishmaniosis, infected cats can represent an additional domestic reservoir for L. infantum to humans, dogs, and other susceptible hosts, including cats themselves. However, there is some evidence that cats are likely less infectious to sand flies than dogs.
Non-vectorial transmission of L. infantum (blood transfusion, vertical transmission, mating, aggressive interactions) is documented in dogs but not yet in cats. However, Leishmania DNA has been detected by PCR in the blood of some healthy cats (see “Diagnosis” and “Prevention” sections).